FARUM, DENMARK, April 17, 2026 /EINPresswire.com/ — MagVenture, a global provider of non-invasive neuromodulation solutions, today marks the publication of a landmark randomized clinical trial evaluating Magnetic Seizure Therapy (MST) for the treatment of Major Depressive Disorder (MDD) in The Lancet Psychiatry.
This milestone represents an important moment for both MagVenture and the broader field of brain stimulation, as the largest Magnetic Seizure Therapy study to date is now available in one of the world’s most prestigious medical journals.
The Canada- and U.S.-based multicenter trial, Confirmatory efficacy and safety trial of magnetic seizure therapy versus right unilateral ultra-brief electroconvulsive therapy in depression (CREST–MST): a randomised, double-blind, non-inferiority trial in Canada and the USA, included 236 participants, making it the most comprehensive MST study conducted to date.
The Phase III study also represents the largest clinical research investment in MagVenture’s history, reflecting the company’s long-term commitment to advancing innovation in convulsive therapies.
Results from the randomized, double-blind, non-inferiority trial demonstrated that MST achieved non-inferior efficacy compared to right unilateral ultra-brief pulse-width electroconvulsive therapy (RUL-UB ECT) in achieving remission of depressive symptoms. At the same time, MST showed a more favorable cognitive safety profile, with substantially fewer patients experiencing clinically meaningful worsening in autobiographical memory.
Additional findings demonstrated comparable response and remission rates across key depression endpoints, similar improvements in suicidality outcomes, time to reorientation, and an overall favorable tolerability profile, with the majority of the reported adverse events classified as mild.
MST is an emerging form of convulsive therapy that has been investigated for its potential antidepressant effects by inducing a therapeutic seizure.
However, MST differs from ECT as it uses a magnetic field to induce a therapeutic seizure. This approach enables more focal brain activation and has been associated with reduced cognitive adverse effects across multiple domains, including memory, attention, and executive function.
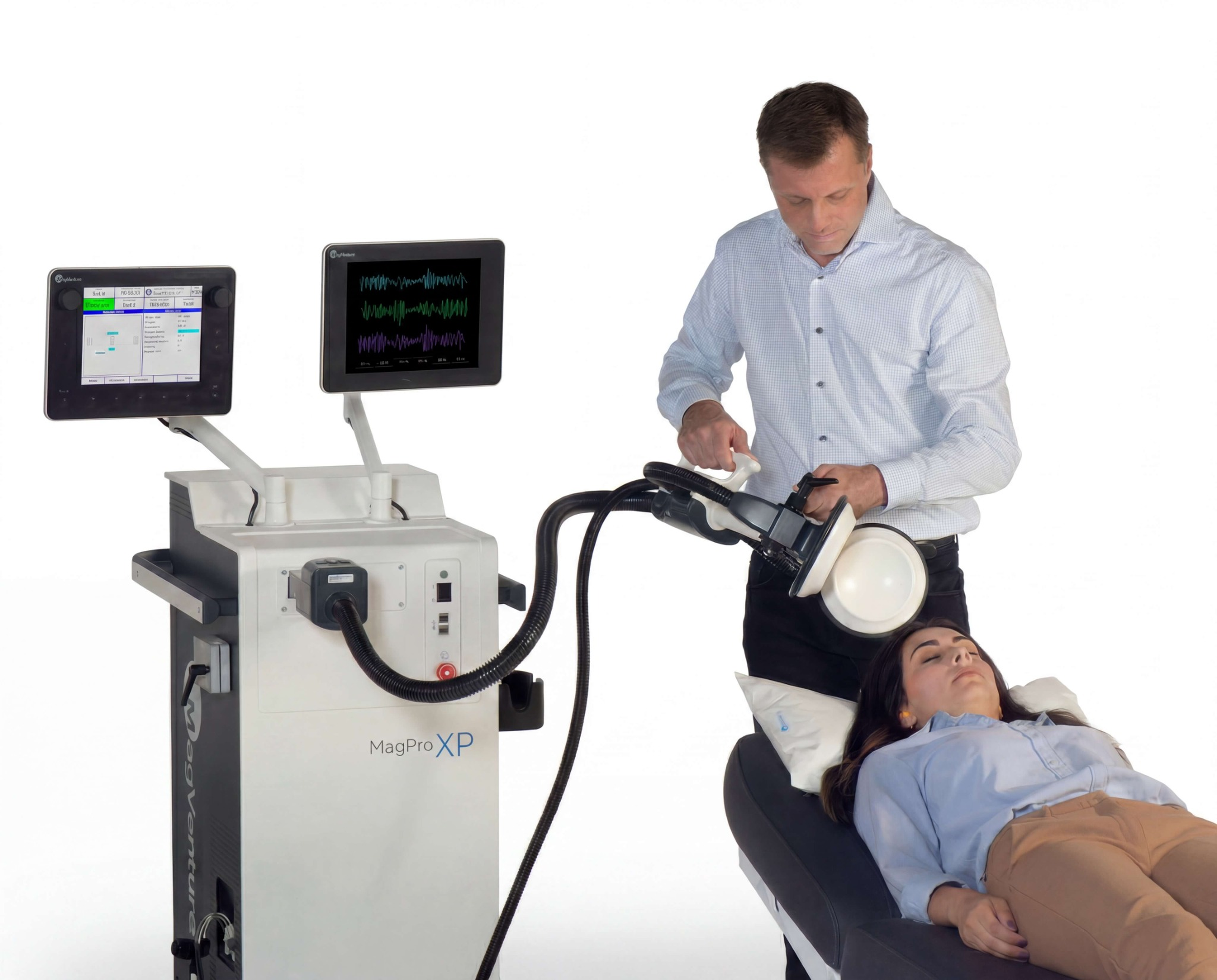
The CREST-MST study was conducted using MagVenture’s proprietary MST technology, including the MagPro XP Black™ magnetic stimulator and the MagVenture TwinCoil™, designed to enable powerful, consistent and controlled delivery of therapeutic stimulation.
The study reflects more than eight years of dedicated research since its initiation in 2018. MagVenture supported the trial by providing MST systems and backup devices across all participating sites, contributing to the successful execution of this large-scale investigation. MagVenture has been engaged in MST research since the early 2000s and remains among the first manufacturers actively developing this technology.
“We are highly encouraged by the publication of this important study in prestigious The Lancet Psychiatry,” said Christian Wanding, CCO of MagVenture A/S. “MagVenture has been deeply involved in the development of MST for more than two decades, and this study represents a significant milestone not only for our organization but for the field as a whole. Supporting this level of rigorous clinical research reflects our long-standing commitment to advancing new treatment modalities that can meaningfully improve patient outcomes, particularly in areas where unmet needs remain high.”
“This study marks a turning point in the evolution of convulsive therapies. MST has the potential to to transform how we treat severe depression by preserving the therapeutic benefits of ECT while significantly improving the patient experience, particularly in terms of cognitive outcomes.” says Dr. Daniel Blumberger, Senior Scientist at CAMH and co-lead of the study.
The results also highlight MST’s potential role in addressing a significant unmet need in depression care, as many patients decline ECT despite its established efficacy due to concerns about cognitive side effects. The CREST-MST findings support MST as a compelling future therapeutic alternative within convulsive therapies, offering a differentiated balance of efficacy and tolerability.
MagVenture is actively pursuing regulatory clearance of MST in multiple markets worldwide and continues to support additional research initiatives across Canada, the United States, Brazil, China, Japan, and Europe, reflecting sustained global interest in this emerging therapeutic approach.
About MagVenture
MagVenture is a Danish-based family-owned medical device company focused on developing and manufacturing non-invasive magnetic stimulation systems used globally in psychiatry, neurophysiology, neurology, cognitive neuroscience, rehabilitation, and pain management. With 35+ years of dedication to magnetic stimulation, MagVenture is committed to delivering technologies that support clinicians in improving patient outcomes across a range of neurological and psychiatric conditions.
Disclaimer
MagVenture Magnetic Seizure Therapy (MST) Systems are not approved or cleared for clinical use in any market worldwide. CAUTION, INVESTIGATIONAL DEVICE. The MagVenture MST System is limited by Federal (or United States) law to investigational use.
Lilja Gudmundsdottir Astrup
MagVenture
+45 42 92 46 06
lga@magventure.com
Visit us on social media:
LinkedIn
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
![]()
Media gallery